

A Collaborative Seminar by GHAIR × AIHS (AI in Healthcare Society), York University, Canada
On 13 February 2026, GHAIR (Global House of Australian Independent Research) and AIHS (AI in Healthcare Society), a registered club of York University, Canada, jointly delivered an interdisciplinary seminar exploring how Artificial Intelligence is transforming brain-cancer research through computational modeling. The seminar was led by Tamour Zubair and Syeda Alishwa Zanib from Global House of Australian Independent Research (GHAIR), who served as the main speakers and delivered a comprehensive presentation on neuro–glioma interactions and the emerging role of AI in predictive biomedical modeling.
We extend our sincere appreciation to Alessia D’Addario and Eisha Rehman, dedicated undergraduate students at York University and active members of AIHS, for their meaningful collaboration and coordination in making this academic exchange possible. Their initiative, enthusiasm, and commitment to interdisciplinary research played an important role in strengthening the partnership between AIHS and GHAIR, reflecting the spirit of global student-led innovation and collaboration.

The seminar brought together neuroscience, mathematical modeling, and artificial intelligence into a unified framework aimed at understanding one of the most complex systems in healthcare: neuro–glioma interaction.
- Research Background:
As illustrated in the figures, neurons communicate through electrical impulses and neurotransmitters. Glial cells such as astrocytes and oligodendrocytes support neuronal function. Under normal conditions, this communication maintains brain stability. However, when glioma develops, this balance is disrupted.
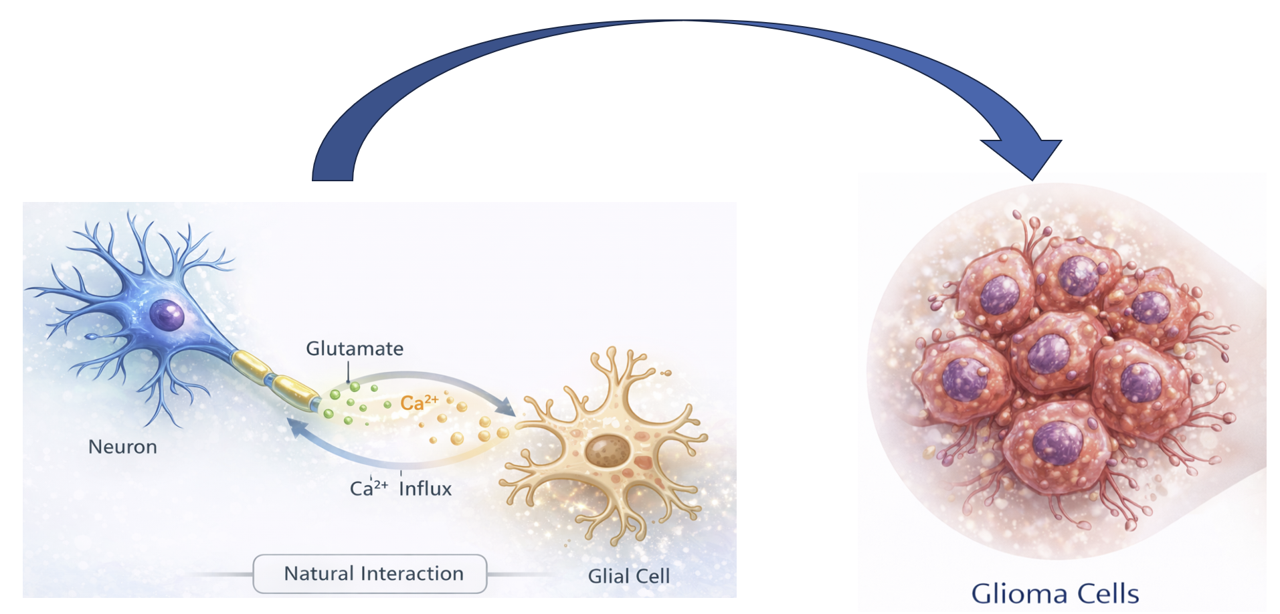
- Neuro-Glioma Cancer: When Signaling Becomes Pathological:
The Neuro-Glioma Cancer figure illustrated the neuron-glioma peritumoral signaling loop, highlighting a key scientific clarification emphasized during the seminar: neurons do not initiate glioma; however, once glioma is established, neuronal signaling actively promotes tumor progression. The diagram demonstrated how glutamate released from neurons activates AMPA and NMDA receptors on glioma cells, triggering calcium (Ca²⁺) influx and stimulating proliferative pathways. It further showed the involvement of TNFR1 and TNFR2 signaling mechanisms, as well as calcium-driven tumor growth processes that facilitate glioma expansion and invasion. As glioma cells integrate into surrounding neural networks, a nonlinear feedback system emerges in which tumor growth and neural activity become dynamically interconnected. Understanding this highly complex and interconnected biological system requires advanced computational and mathematical modeling approaches capable of capturing its nonlinear, multi-scale behavior.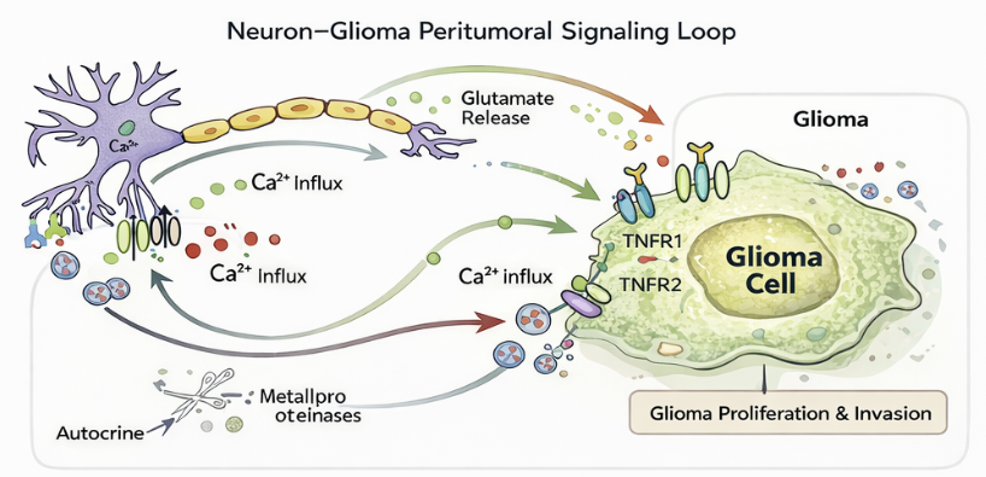
- From Biology to Mathematics Language:
The transition from biological understanding to quantitative modeling represents a crucial step in modern neuro-oncology research. In our framework, the progression follows a structured pathway: Biology → Mathematics → Computation & AI. Biological mechanisms underlying neuron–glioma interactions are translated into nonlinear differential equations that capture neuronal voltage dynamics, calcium concentration fluctuations, tumor growth evolution, and network-level interactions. Mathematical modeling provides a structured way to simulate and analyze how these complex systems evolve over time. However, because neuro–glioma systems are inherently high-dimensional, nonlinear, and dynamically interconnected, mathematical modeling alone is not sufficient—advanced computational techniques and artificial intelligence are required to manage complexity, optimize parameters, and enhance predictive capability.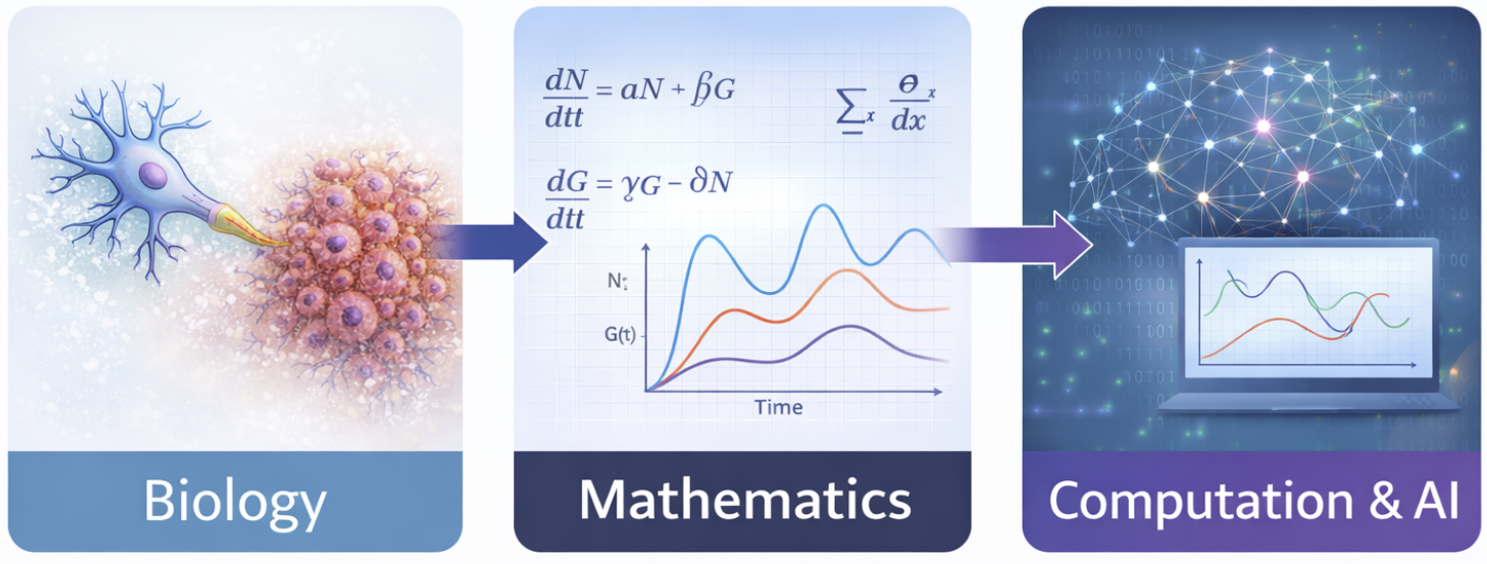
- The Role of Artificial Intelligence:
Artificial intelligence plays a transformative role in enhancing modern research workflows by enabling efficient parameter optimization, supporting experimental and simulation design, improving code efficiency to reduce computational cost, facilitating advanced data handling and pattern discovery, strengthening model development and refinement, ensuring reproducible research processes, and even assisting in manuscript preparation and presentation development. By integrating AI into computational frameworks, researchers are better equipped to manage large and high-dimensional datasets, uncover hidden nonlinear interactions within complex biological systems, and significantly improve predictive accuracy and analytical robustness.
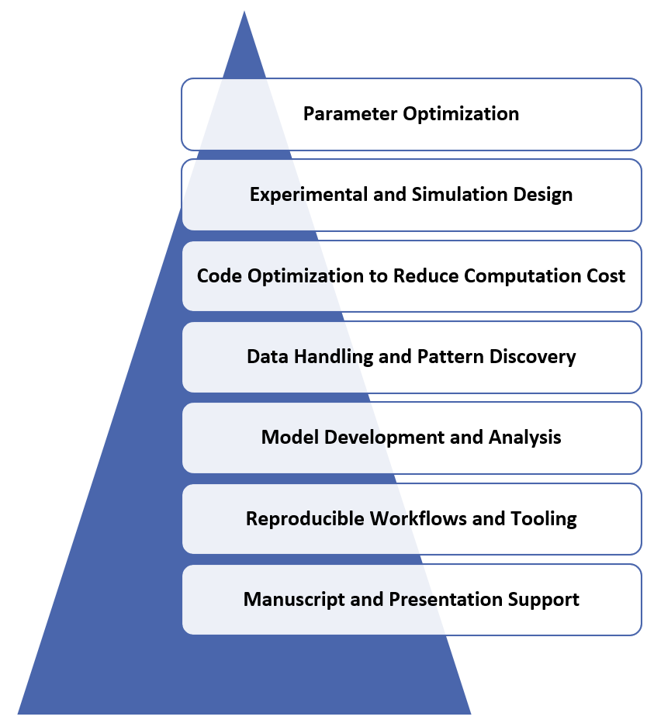
- Why AI Matters in Brain & Cancer Studies:
Brain–cancer systems are among the most complex biological environments in healthcare research. Neuro–glioma interactions involve dynamic calcium signaling, electrical neuronal activity, tumor proliferation, and multi-scale cellular communication occurring simultaneously. These systems generate large, high-dimensional datasets with nonlinear and often hidden interactions that are difficult to interpret using traditional analytical methods alone. Artificial Intelligence provides the computational power and analytical flexibility needed to model such complexity. By identifying hidden patterns, managing multidimensional data, and enhancing predictive modeling, AI enables researchers to move beyond descriptive understanding toward data-driven, optimized, and clinically relevant insights. In the context of neuro-oncology, AI is not just a supportive tool — it is a transformative engine for advancing precision research.
